ShmoopTube
Where Monty Python meets your 10th grade teacher.
Search Thousands of Shmoop Videos
Problem Solving and Data Analysis Videos
A mole is a unit used in chemistry to measure atoms, molecules, electrons, and other particles in a given amount of substance. We know that the molar mass of mercury is approximately 200 grams per mole. Using a small calibrated test tube containing water, a scientist is able to determine that the volume of a small amount of mercury that he will use in his experiment is 24.5 cubic centimeters. If mercury has a density of 13.5 grams per cubic centimeter, how many moles of mercury are in 24.5 cubic centimeters? Round your answer to one decimal place.
Center, shape, and spread Videos 3 videos
Employees of a candy company surveyed people of different ages and asked them their favorite flavor of candy. One of the surveyors chooses to send...
Data collection and evaluation Videos 6 videos
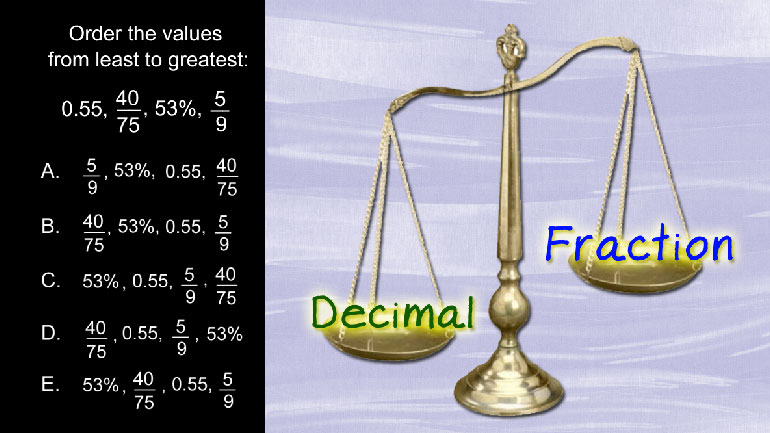
SAT Math 1.1 Statistics and Probability. In which of the following data sets are the arithmetic mean and the median equal?
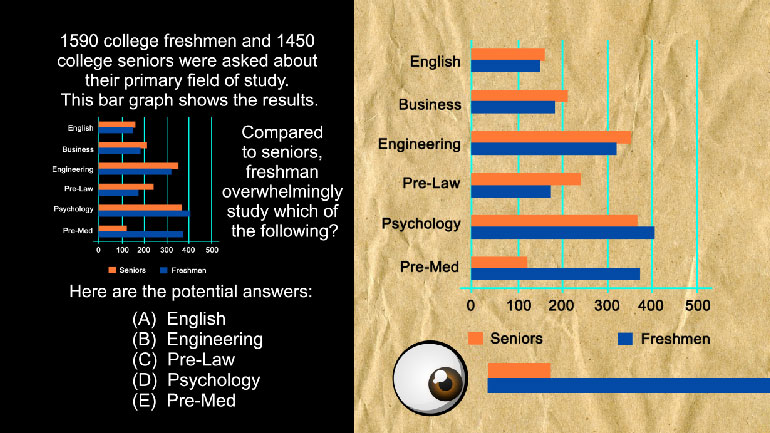
Data inferences Videos 7 videos
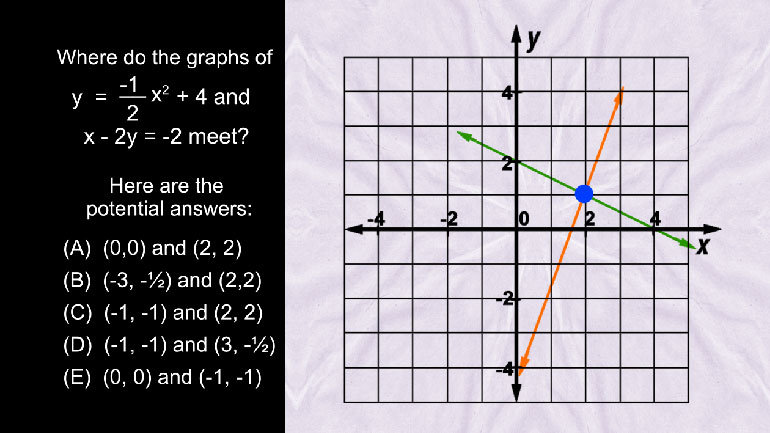
SAT Math 1.1 Algebra and Functions. Find an algebraic equation to correspond with the data.
SAT Math 2.2 Numbers and Operations. According to the information given, how many times faster is Car A than Car D?
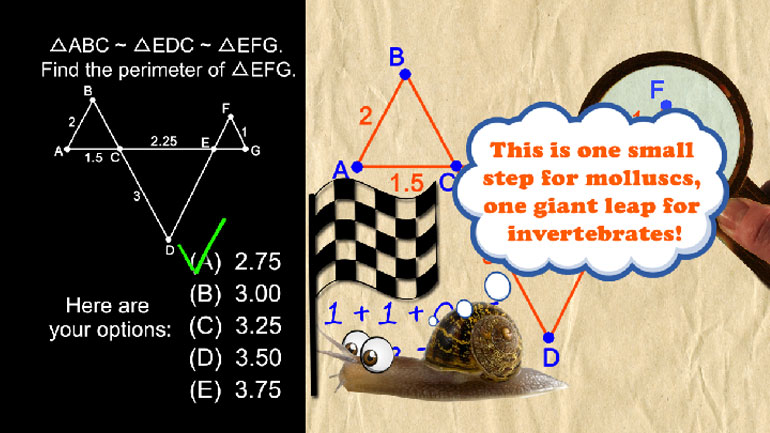
Key features of graphs Videos 10 videos
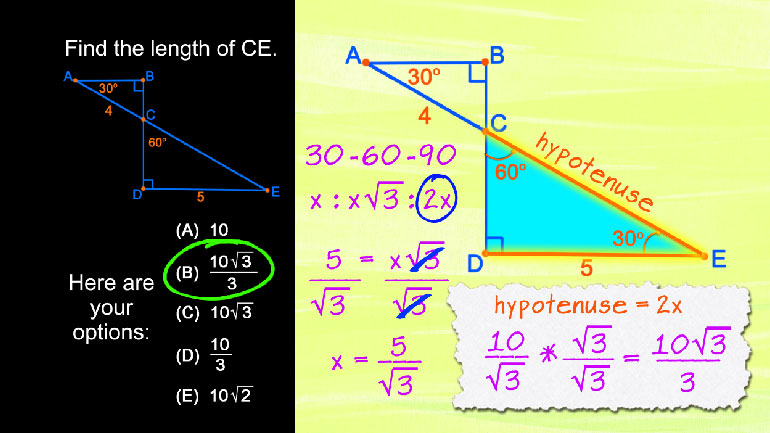
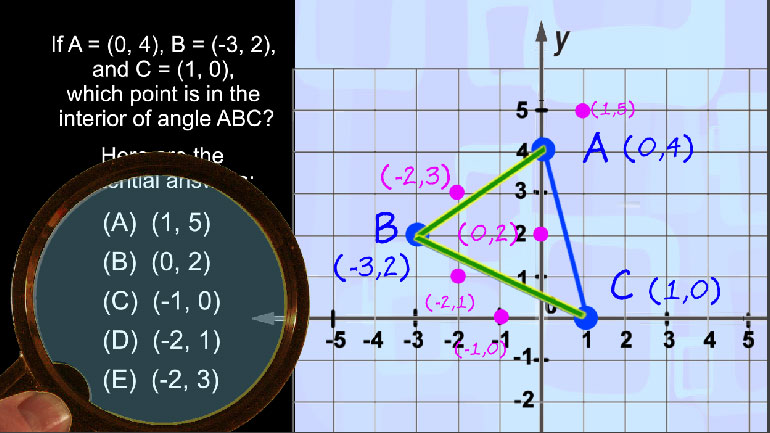
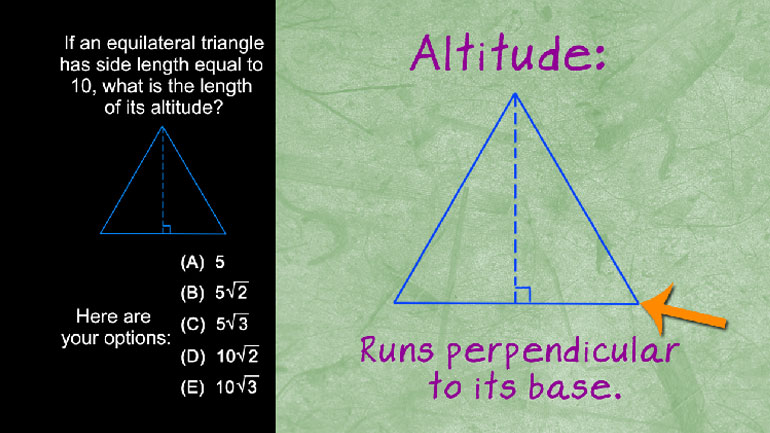
SAT Math 1.3 Geometry and Measurement. Find the length of CE.
Percentages Videos 6 videos
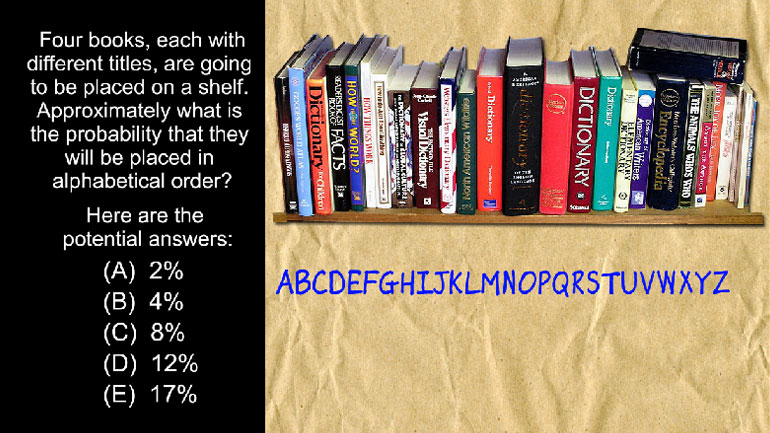
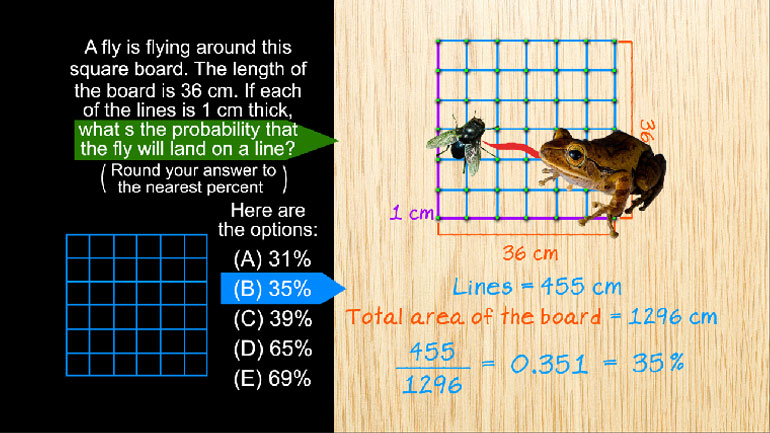
SAT Math 1.3 Statistics and Probability. Approximately what is the probability that they will be placed in alphabetical order?
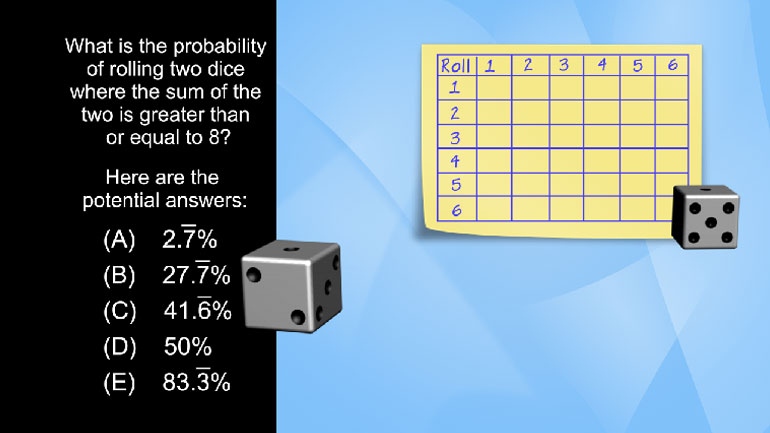
SAT Math 1.5 Statistics and Probability. If Silas draws one card, then places it in his pocket and draws another, what is the probability that...
Ratios, rates, and proportions Videos 10 videos
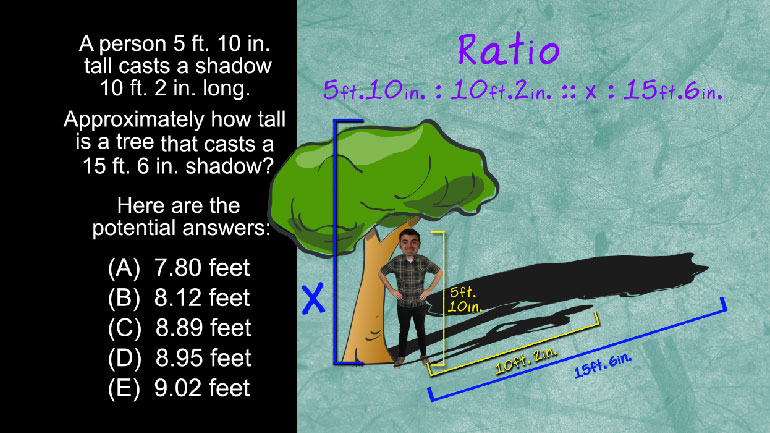
SAT Math 10.3 Geometry and Measurement. What is the ratio of birds to dogs?
Two-way table data Videos 7 videos
SAT Math: Algebra and Functions Drill 1, Problem 2. Based on the data in the table, what is the maximum heart rate for the average 22-year-old?