ShmoopTube
Where Monty Python meets your 10th grade teacher.
Search Thousands of Shmoop Videos
Heart of Algebra Videos
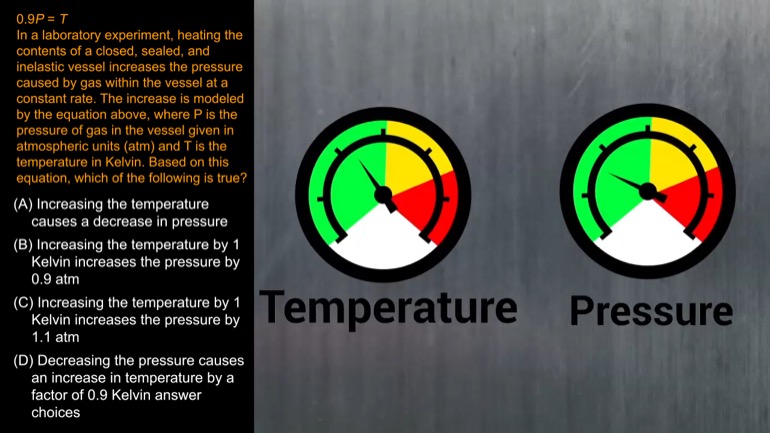
In a laboratory experiment, heating the contents of a closed, sealed, and inelastic vessel increases the pressure caused by gas within the vessel at a constant rate. The increase is modeled by the equation above, where P is the pressure of gas in the vessel given in atmospheric units (atm) and T is the temperature in Kelvin. Based on this equation, which of the following is true?
Interpreting linear functions Videos 4 videos
SAT Math 1.5 Numbers and Operations. How many dots would be in the 5th term of this sequence?
SAT Math 2.3 Numbers and Operations. If Joaquin invests $500, how much will the investment be worth after 3 years?
Linear equations in one variable (word problems) Videos 6 videos
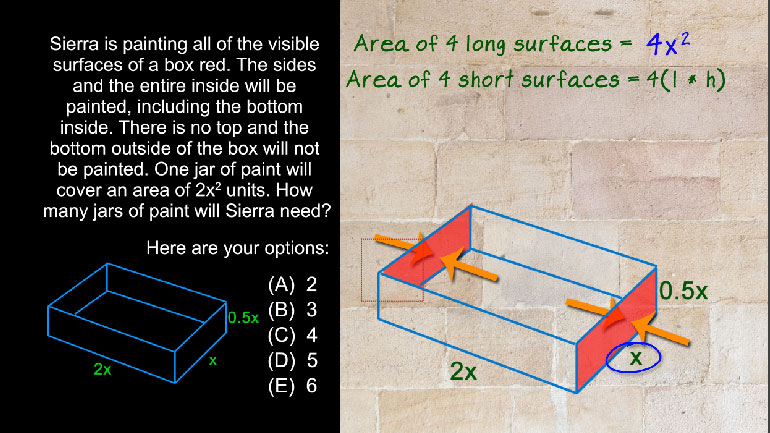
SAT Math 1.5 Geometry and Measurement. How many jars of paint will Sierra need?